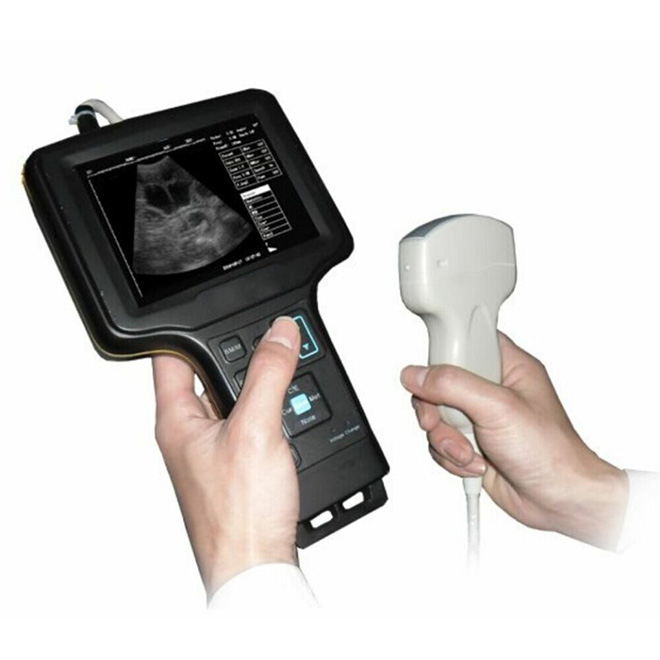
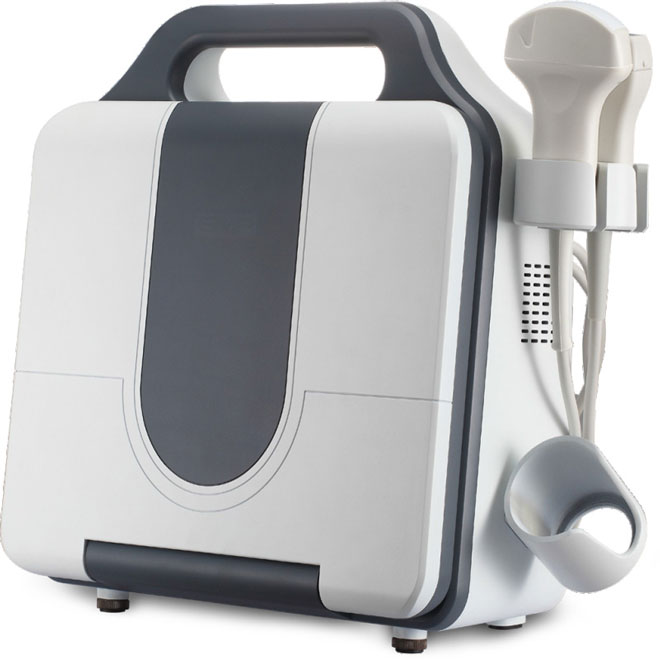
Portable Bone Density Scanner MDLBD09
Non-invasive quantitative ultrasound system for rapid osteoporosis screening and bone density assessment. Portable design with critical angle side wave technology enables safe, radiation-free measurement of radius and tibia bone density across all patient populations including pregnant women, children, and elderly patients.






Clinical overview
The MDLBD09 is a portable, non-invasive quantitative ultrasound (QUS) system designed for rapid osteoporosis screening and bone density assessment. Using critical angle side wave theory, the device measures bone density at peripheral sites (radius or tibia) and provides comprehensive bone health parameters including speed of sound (SOS), T-score, Z-score, skeletal age, bone quality index (BQI), relative risk factor (RRF), and expected osteoporotic age (EOA). Single measurements complete in ≤25 seconds, enabling high-throughput screening in clinical settings.
The system is particularly valuable for populations where DXA scanning is impractical or contraindicated: pregnant women, children, elderly patients in nursing facilities, and mobile screening programs. Measurement results are unaffected by soft tissue thickness, bone size, or skeletal shape variations, ensuring reliable assessment across diverse body types. Multi-region reference databases (Asia, Europe, and other regions) with multi-age data enable accurate interpretation across diverse demographic populations. Windows XP or higher compatibility allows integration with desktop computers, laptops, or tablets for flexible clinical deployment.
Key features & surgeon benefits
Radiation-Free Technology
- Zero radiation exposure to patients and staff
- Safe for pregnant women and pediatric populations
- Suitable for frequent serial measurements
- No radiation safety infrastructure required
Rapid Point-of-Care Testing
- Single measurement in ≤25 seconds
- Repeat measurements in ≤75 seconds
- Immediate results for same-visit counseling
- High patient throughput capability
Cost-Effective Screening
- Lower capital investment than DXA systems
- Reduced operating costs and maintenance
- No specialized facility modifications needed
- Accessible for smaller practices and screening programs
Lightweight Portable Design
- Easy transportation between multiple locations
- Suitable for mobile screening programs
- Enables nursing home and community health screenings
- Minimal space requirements in clinical settings
Technical specifications
| Indications | Osteoporosis screening, bone density assessment, fracture risk evaluation, pediatric bone development monitoring, postmenopausal women assessment, elderly population screening |
|---|---|
| Measurement sites | Radius or tibia (peripheral bone sites) |
| Measurement technology | Critical Angle Side Wave Theory with quantitative ultrasound |
| Probe frequency | 1 MHz ±15% |
| Measurement parameters | SOS (Speed of Sound), T-score, Z-score, Skeletal Age, BQI (Bone Quality Index), RRF (Relative Risk Factor), EOA (Expected Osteoporotic Age) |
| Single measurement time | ≤25 seconds |
| Repetition time | ≤75 seconds between measurements |
| Operating system | Windows XP or higher |
| Reference database | Multi-age data from Asia, Europe, and other regions |
| Soft tissue independence | Results not affected by soft tissue thickness, bone size, or skeletal shape |
| Operating temperature | 5°C to 40°C |
| Operating humidity | ≤80% relative humidity |
| Power requirement | AC 220V ±10%, 50Hz ±1Hz |
| Design | Portable and lightweight for easy transportation |
| Warranty | 2 Year Manufacturer Warranty |
Standard instrument composition
| Item | Description | Qty |
|---|---|---|
| Main unit | Portable bone density scanner with integrated ultrasound probe | 1 |
| Ultrasound probe | 1 MHz ±15% critical angle side wave transducer | 1 |
| Coupling gel | Medical-grade ultrasound coupling medium | As applicable |
| Software | Windows-based measurement and analysis software with multi-region database | 1 license |
| Power cable | AC 220V ±10%, 50Hz ±1Hz connection | 1 |
| User manual | Comprehensive operating and maintenance documentation | 1 |
| Quality control phantom | Calibration and quality assurance device | 1 |
Get in touch for pricing & availability
Contact our sales team to discuss bone density screening solutions for your practice. We provide comprehensive support including installation, operator training, and ongoing technical assistance.
Email: info@medicaldevicelab.com
Hours: Monday–Friday, 8 AM–6 PM PST | 24/7 Emergency Support Available








Reviews
There are no reviews yet.