TLC6000 Dynamic ECG Systems
Advanced 12-lead ECG recording and analysis system for continuous cardiac monitoring up to 48 hours. Delivers comprehensive arrhythmia detection, pacemaker analysis, and predictive risk assessment with intuitive PC-based software integration for clinical and community settings.



Clinical overview
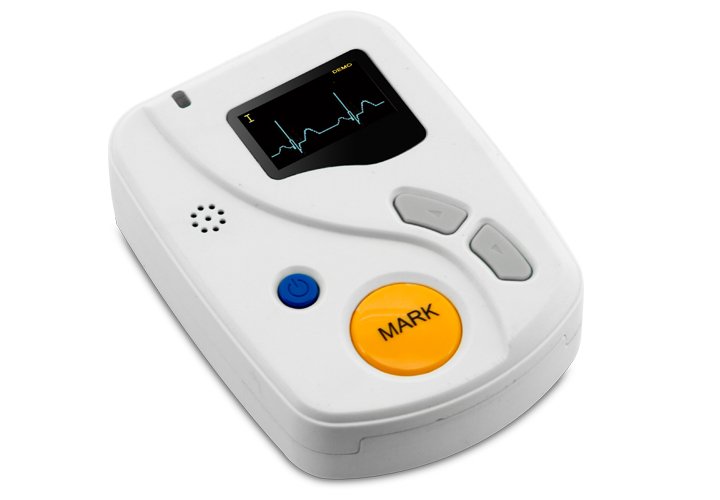
The TLC6000 Dynamic ECG System represents a comprehensive solution for continuous cardiac monitoring and arrhythmia detection in medical institutions and community healthcare settings. Adopting the international 12-lead standard, this system enables uninterrupted ECG waveform recording for up to 48 hours with synchronized multi-lead analysis. The integrated PC software platform provides clinicians with advanced diagnostic capabilities including automated arrhythmia classification, pacemaker function assessment, and predictive risk stratification for myocardial infarction and sudden cardiac death.
With over 10 pre-defined pathologic waveform templates and flexible user-defined modules, the TLC6000 delivers precise QRS identification without distortion, ensuring diagnostic accuracy across diverse patient populations. The system’s powerful analysis engine supports all major pacemaker types (AAI, VVI, DDD, etc.) and provides comprehensive heart rate variability assessment across multiple time intervals, making it an essential tool for both acute and long-term cardiac evaluation.
Key features & surgeon benefits
Comprehensive Arrhythmia Detection
- 12-lead international standard system for complete cardiac coverage
- Over 10 pre-defined templates for atrial fibrillation, premature beats, and pathologic waveforms
- User-defined modules for rare or institution-specific arrhythmia patterns
Streamlined Analysis & Reporting
- Fast review analysis for single or full-lead ECG in any time bucket
- One-key report printing for rapid clinical decision-making
- Synchronized multi-lead analysis with selectable main analysis channel
Predictive Risk Assessment
- HRT (Heart Rate Turbulence) analysis for myocardial infarction mortality prediction
- T-Wave alternation detection for malignant arrhythmia and sudden cardiac death risk
- Heart rate variability analysis at 5-minute, 1-hour, and whole-recording intervals
Specialized Diagnostic Modules
- Comprehensive pacemaker analysis for all major device types
- QTD (QT dispersion) and VCG (vector cardiogram) assessment
- VLP (ventricular late potential) and TVCG (time vector cardiogram) analysis
Technical specifications
| Indications | Cardiac arrhythmia detection, myocardial infarction risk assessment, pacemaker function analysis, heart rate variability evaluation, sudden cardiac death risk stratification |
|---|---|
| Sizes / range | 12-lead system; continuous ECG recording up to 48 hours |
| Material | Medical-grade electronic components |
| Sterilisation | As applicable |
| Packaging | Standard clinical packaging with accessories |
| Warranty | 2 Year Manufacturer Warranty |
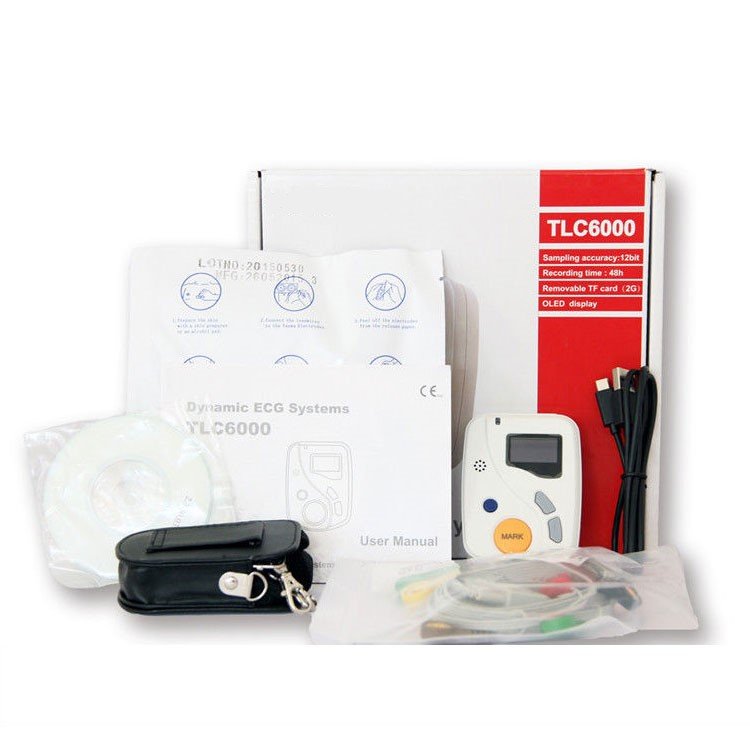
Standard instrument composition
| Item | Description | Qty |
|---|---|---|
| ECG Recording Unit | 12-lead portable recorder with 48-hour continuous recording capability | 1 |
| PC Analysis Software | Comprehensive diagnostic software with multi-lead synchronization and automated analysis modules | 1 |
| Lead Set | Complete 12-lead electrode configuration with international standard connectors | 1 |
| Electrode Pads | Medical-grade adhesive electrodes for patient application | As required |
| Data Cable | USB/serial interface for ECG unit to PC connection | 1 |
| Power Supply | AC adapter and rechargeable battery pack | 1 |
| User Documentation | Clinical manual, software guide, and quick-reference cards | 1 set |
Get in touch for pricing & availability
Contact our clinical sales team to discuss system configuration, institutional pricing, and integration with your existing cardiac monitoring infrastructure. We provide comprehensive support and training for seamless implementation.
Email: info@medicaldevicelab.com















Reviews
There are no reviews yet.