Portable Bone Density Scanner MDLBD01
Ultra-portable quantitative ultrasound bone densitometer weighing less than 4 kg. Delivers dual-emission precision technology for radius and tibia assessment with comprehensive T-Score, Z-Score, and Bone Quality Index analysis. Radiation-free, dry measurement technology with advanced DuPont probe and EMC certification for reliable point-of-care osteoporosis screening.
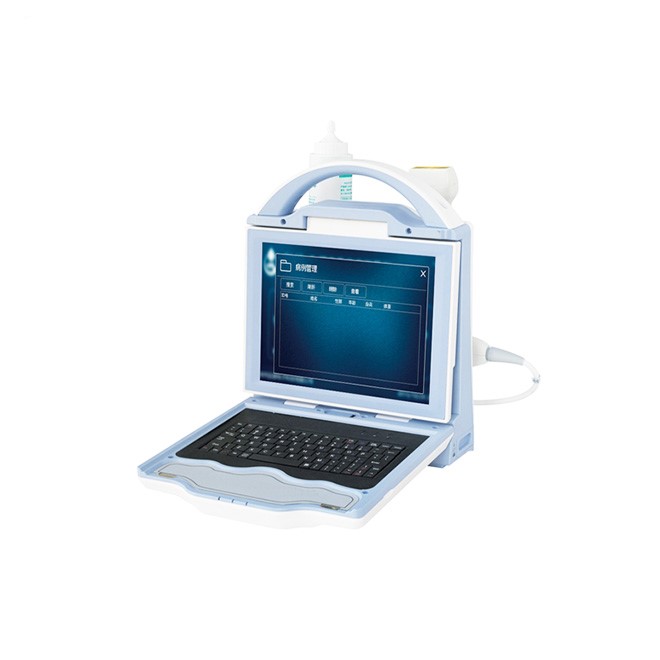


Clinical overview
The MDLBD01 represents a breakthrough in accessible bone health assessment, combining hospital-grade diagnostic capabilities with unprecedented portability. This quantitative ultrasound (QUS) bone densitometer measures bone density at the radius and tibia—clinically relevant peripheral sites that predict osteoporotic fracture risk across all skeletal regions. Unlike traditional DXA systems requiring dedicated rooms and radiation shielding, the MDLBD01 operates anywhere with a power outlet, enabling point-of-care screening in primary care clinics, nursing homes, community health programs, and mobile health units.
The dual-emission and dual-reception ultrasound technology enhances measurement accuracy by analyzing bone density from multiple angles simultaneously, compensating for soft tissue variations and providing results comparable to central DXA for peripheral sites. Advanced algorithms minimize measurement errors while comprehensive reference data spanning multiple ethnicities and age ranges (0–100 years) ensures diagnostic accuracy across diverse patient populations. The fully dry measurement technology eliminates coupling gel, reducing examination time, improving patient comfort, and simplifying infection control protocols. With radiation-free assessment, rapid measurement time (<1 minute per site), and internet-enabled remote service capabilities, the MDLBD01 is ideally suited for frequent monitoring, pediatric applications, and longitudinal treatment response evaluation.
Key features & surgeon benefits
Ultra-Portable Design
- Weighs less than 4 kg for true point-of-care deployment
- Eliminates need for dedicated DXA rooms and radiation shielding
- Enables screening in clinics, nursing homes, and community settings
Dual-Emission Precision
- Dual-emission and dual-reception technology for enhanced accuracy
- Analyzes bone density from multiple angles simultaneously
- Compensates for soft tissue variations and positioning errors
Dry Measurement Technology
- Fully dry diagnostic operation eliminates coupling gel
- Reduces examination time and improves patient comfort
- Simplifies probe disinfection and eliminates gel consumable costs
Radiation-Free Assessment
- Ultrasound-based technology provides zero radiation exposure
- Safe for frequent monitoring, pediatric patients, and pregnant women
- Enables longitudinal treatment monitoring without cumulative radiation risk
High-Definition Visualization
- 10.4-inch LED color monitor for clear result visualization
- Intuitive graphical interface displays T-scores, Z-scores, and fracture risk
- Adjustable viewing angle accommodates various clinical settings
Comprehensive Analysis Metrics
- T-Score, Z-Score, Age Percentile, Adult Percentile, Bone Quality Index (BQI)
- Physiological Bone Age, Expected Osteoporosis Age, Relative Fracture Risk
- Advanced error correction algorithms ensure diagnostic-grade accuracy
DuPont Advanced Technology
- DuPont ultrasound transducers for superior sensitivity and durability
- IPX7-rated waterproof probe withstands full submersion for disinfection
- Enhanced signal-to-noise ratio ensures consistent measurements
EMC Safety & Compliance
- First device in class to pass rigorous EMC testing
- Ensures reliable operation alongside other medical equipment
- Guarantees no electromagnetic interference with clinical environment
Technical specifications
| Indications | Osteoporosis screening, bone density assessment, fracture risk evaluation, treatment monitoring in patients aged 0–100 years |
|---|---|
| Measurement sites | Radius (distal 1/3 forearm) and Tibia (mid-shaft) |
| Technology | Quantitative Ultrasound (QUS) with dual-emission and dual-reception |
| Display | 10.4-inch high-definition color LED monitor |
| Weight | Less than 4 kg (ultra-portable) |
| Measurement time | Less than 1 minute per site |
| Operating mode | Dry measurement (no coupling gel required) |
| Probe technology | DuPont advanced ultrasound transducers with superior sensitivity |
| Waterproof rating | Main unit: IPX0; Probe: IPX7 (fully submersible) |
| Power supply | Medical-grade external power adapter (>60W) |
| Connectivity | Internet-enabled for remote service, updates, and data access |
| EMC certification | First-in-class EMC test certified device |
| Reference database | Multi-ethnic (European, American, Asian, Chinese) plus WHO standards |
| User interface | English language menu system |
| Data storage | Internal memory with patient database management |
| Warranty | 2 Year Manufacturer Warranty |
Measurement parameters & analysis metrics
| Parameter | Description |
|---|---|
| T-Score | Comparison to peak bone mass of healthy young adults (standard deviation units); diagnostic threshold for osteoporosis |
| Z-Score | Comparison to age-matched reference population (standard deviation units); identifies abnormal bone density for age |
| Age Percentile [%] | Patient’s bone density ranking within same age group; contextualizes results relative to peers |
| Adult Percentile [%] | Patient’s bone density ranking within adult reference population; identifies deviation from adult norms |
| Bone Quality Index (BQI) | Comprehensive bone health assessment combining multiple ultrasound parameters; reflects both density and microarchitecture |
| Physiological Bone Age (PAB) | Estimated skeletal age based on bone density measurements; identifies premature or delayed skeletal aging |
| Expected Osteoporosis Age (EOA) | Projected age at which osteoporosis threshold may be reached; enables preventive intervention planning |
| Relative Fracture Risk (RRF) | Quantified fracture risk assessment compared to reference population; guides treatment decisions |
Standard instrument composition
| Item | Description | Qty |
|---|---|---|
| Main Unit | MDLBD01 portable bone density scanner with 10.4″ LED display, dual-emission/dual-reception ultrasound system, internal memory, and internet connectivity | 1 |
| Ultrasound Probe | DuPont advanced transducer assembly; IPX7-rated waterproof; dry coupling technology; includes probe cable and connector | 1 |
| Power Adapter | Medical-grade external power supply (>60W output); includes power cable and connector | 1 |
| Probe Stand | Protective stand for probe storage and positioning during operation | 1 |
| Quality Control Phantom | Calibration phantom for weekly quality assurance measurements | 1 |
| User Manual | Comprehensive operating instructions, clinical guidelines, and troubleshooting documentation | 1 |
| Quick Reference Guide | Laminated clinical reference card with measurement sites, patient positioning, and result interpretation | 1 |
| Carrying Case | Protective transport case for portable deployment and storage | 1 |
Get in touch for pricing & availability
Contact our sales team to discuss pricing, lease options, and deployment strategies for your clinical setting. We provide comprehensive support from installation through ongoing technical service and software updates.
Email: info@medicaldevicelab.com
Hours: Monday–Friday, 8 AM–6 PM PST; 24/7 Emergency Support Available










Reviews
There are no reviews yet.