Handheld Portable ETCO2 Capnograph Monitor CA10M
Compact, handheld capnograph for real-time end-tidal CO2 and respiratory rate measurement. Designed for pre-hospital emergency, ICU, and operating room use across adult, pediatric, and neonatal populations with high-precision monitoring and intuitive alarms.
Clinical overview
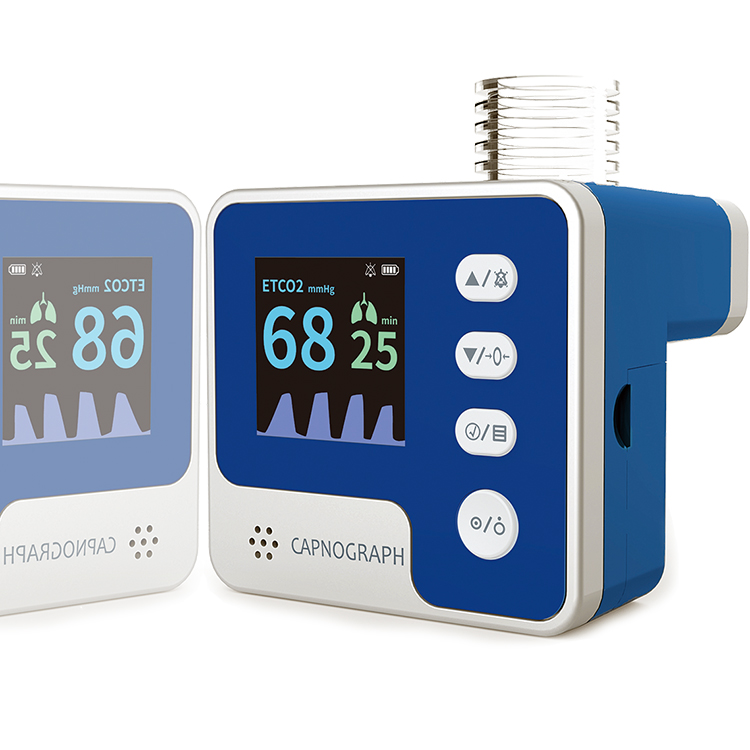
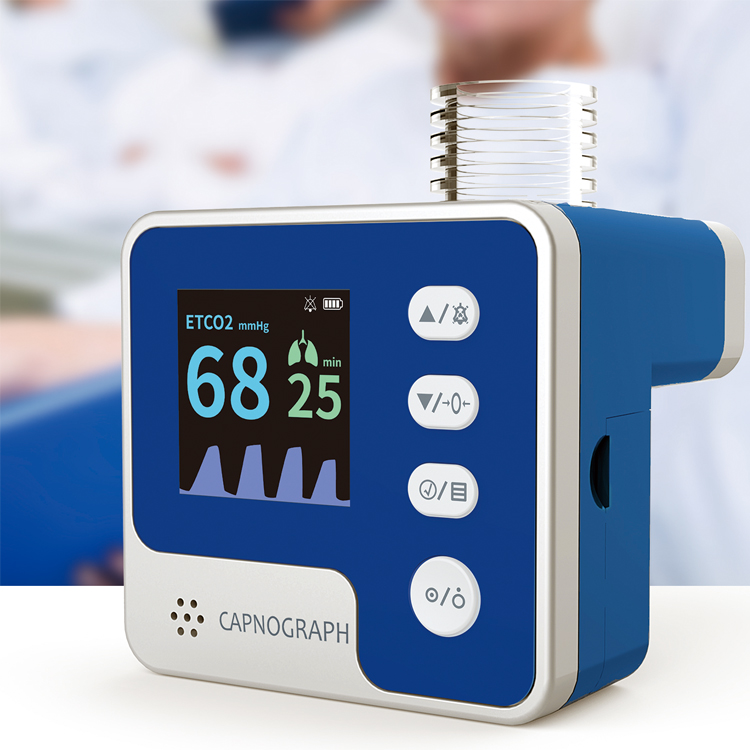
The CA10M capnograph employs mainstream capnography methodology to measure end-tidal carbon dioxide (ETCO2), airway respiration rate, and related parameters. By detecting pulmonary ventilation function, it reflects circulation and pulmonary blood flow conditions, while indirectly assessing alveolar ventilation. This makes it ideal for pre-hospital emergency transfer, emergency departments, ICUs, operating rooms, neonatology units, and respiratory departments.
The device connects to the patient’s respiratory tract via an airway adapter and breathing tube, providing continuous, non-invasive monitoring across adult, pediatric, and neonatal populations. Its compact form factor (55×52×59 mm, 97 g with batteries) and user-friendly display enable rapid deployment in time-critical settings without compromising measurement accuracy or clinical reliability.
Key features & surgeon benefits
Precise ETCO2 & RR Detection
- CO2 range 0–150 mmHg with 1 mmHg resolution
- Respiration rate 2–150 rpm with ±1 rpm accuracy
- Tiered accuracy: ±2 mmHg (0–40), ±5% (41–70), ±8% (71–100), ±10% (101–150)
Enhanced Patient Monitoring
- Over-limit alarms for ETCO2 and respiration rate thresholds
- Low battery alarm ensures uninterrupted monitoring
- Type BF applied part; IPX2 protection against liquid ingress
Compact & Lightweight Design
- 55×52×59 mm dimensions fit easily into emergency kits
- 97 g weight (with batteries) for rapid deployment
- DC 3V operation (2 AAA batteries) requires no external power
Multi-Population Support
- Adult, pediatric, and neonatal gas path adapters
- Operates 5–40°C, 30–75% relative humidity
- Suitable for pre-hospital, ICU, OR, and neonatology settings
Technical specifications
| Indications | Pre-hospital emergency, emergency department, ICU, operating room, neonatology, respiratory monitoring |
|---|---|
| Patient Population | Adult, pediatric, and neonatal |
| CO2 Measurement Range | 0–150 mmHg, 1 mmHg resolution |
| CO2 Accuracy | 0–40 mmHg: ±2 mmHg; 41–70: ±5%; 71–100: ±8%; 101–150: ±10% |
| Respiration Rate Range | 2–150 rpm, 1 rpm resolution, ±1 rpm accuracy |
| Physical Dimensions | 55×52×59 mm |
| Weight | 97 g (with batteries) |
| Power Supply | DC 3V (2 AAA dry batteries), ≤1 VA input |
| Operating Temperature | 5–40°C |
| Relative Humidity | 30–75%, no condensation |
| Atmospheric Pressure | 700–1060 hPa |
| Protection Class | Type BF applied part; IPX2 liquid ingress protection |
| Device Type | Internally powered equipment |
| Warranty | 2 Year Manufacturer Warranty |
Standard instrument composition
| Item | Description | Qty |
|---|---|---|
| CA10M Monitor | Handheld ETCO2 capnograph with display and controls | 1 |
| Adult Gas Path Adapter | Mainstream airway adapter for adult patients | 1 |
| Breathing Tube | Connection tubing for respiratory circuit | 1 |
| User Manual | Operating instructions and clinical guidelines | 1 |
| Neonate Gas Path Adapter | Optional adapter for neonatal applications | Optional |
Get in touch for pricing & availability
Contact our clinical sales team to discuss the CA10M capnograph, request a demonstration, or place an order. We provide rapid delivery and comprehensive support for all clinical environments.
Email: info@medicaldevicelab.com














Reviews
There are no reviews yet.