Bone Density Scan MDLBD05
Advanced ultrasonic bone densitometry system for accurate, non-invasive assessment of bone health. Measures BUA, SOS, and OI using dry-type dual-transmission technology with automatic calibration and multi-ethnic reference databases for diverse populations.

Clinical overview
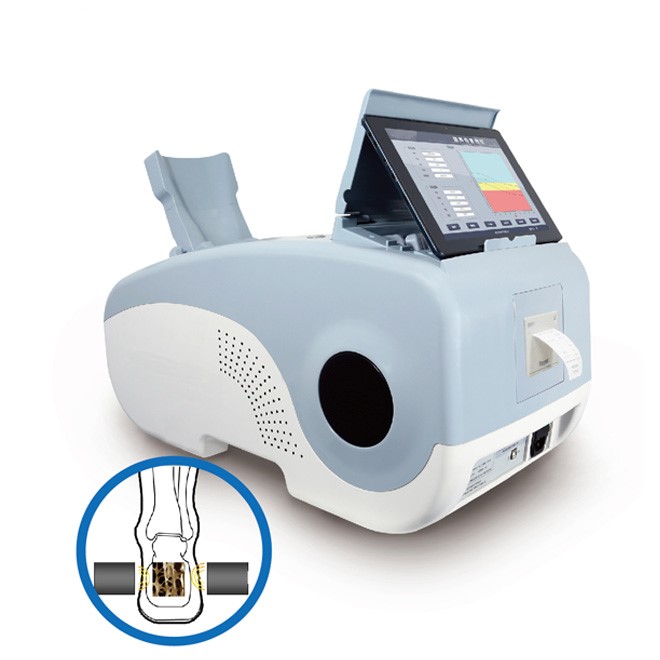
The MDLBD05 represents advanced quantitative ultrasound bone densitometry, delivering professional-grade osteoporosis assessment without ionising radiation. This system measures bone density at the calcaneus (heel bone), a predominantly trabecular site that responds rapidly to aging, disease, and therapeutic interventions. Calcaneal measurements predict hip and vertebral fractures as effectively as central DXA, validating heel bone assessment for clinical fracture risk stratification.
Revolutionary dry-type dual-transmission technology eliminates coupling gel requirements, transforming bone density assessment from a time-consuming procedure into a rapid 25-second scan. The oil bladder probe couples directly to skin without gel application or removal, dramatically improving workflow efficiency in high-volume screening programs. Automatic temperature compensation maintains measurement accuracy across varying environmental conditions, while the permanent probe design eliminates consumable replacement costs and ensures consistent performance throughout the device’s operational life.
Multi-ethnic reference databases (Asian, European, Middle Eastern, African) ensure diagnostic accuracy across diverse patient demographics. Bone density distributions vary significantly between populations—the MDLBD05’s ethnicity-specific normative data prevents systematic over-diagnosis or under-diagnosis from inappropriate reference standards. Comprehensive parameter output (BUA, SOS, OI, T-score, Z-score, adult ratio, age ratio, OPR) provides multidimensional bone health assessment supporting nuanced clinical decision-making and research applications.
Key features & surgeon benefits
Dry-Type Dual-Transmission Ultrasound
- Full dry measurement eliminates coupling gel mess and cleanup time
- Dual-way ultrasonic transmission analyzes bone from multiple propagation paths for enhanced accuracy
- Oil bladder probe provides permanent operational lifespan with no consumable components
- Direct skin coupling accelerates workflow and improves patient comfort
Rapid Assessment & Automatic Reporting
- Complete measurement in ≤25 seconds enables high-throughput screening
- Automatic calibration with human body simulation module ensures consistent accuracy
- Intelligent report generation automatically interprets results and classifies bone density status
- USB, Bluetooth, and PACS connectivity integrate seamlessly with hospital information systems
Eight Comprehensive Parameters
- BUA, SOS, OI, T-score, Z-score, adult ratio, age ratio, OPR provide multidimensional assessment
- Exceptional repeatability (±1% OPR, ±2% SOS, ±5% BUA) enables reliable longitudinal monitoring
- Automatic temperature compensation maintains accuracy across environmental variations
- Detailed parameter analysis identifies compromised bone quality despite normal density
Multi-Ethnic Databases & Customisation
- Built-in reference databases for Asian, European, Middle Eastern, and African populations
- Ethnicity-specific normative data prevents systematic diagnostic errors from inappropriate references
- Comprehensive patient database with automatic case recording and EXCEL export
- Language switching (Chinese/English) and customisable report formats support diverse markets
Technical specifications
| Indications | Osteoporosis screening, bone density monitoring, fracture risk assessment, treatment response evaluation, metabolic bone disease assessment |
|---|---|
| Measurement site | Calcaneus (heel bone) — predominantly trabecular bone |
| Technology | Quantitative Ultrasound (QUS) with dry-type dual-transmission |
| Probe frequency | 0.5 MHz ± 10% |
| Probe design | Oil bladder probe with permanent operational lifespan |
| Measurement time | ≤25 seconds per scan |
| SOS accuracy | ±2% (Speed of Sound) |
| OPR repeatability | ±1% (Osteo Profile Ratio) |
| BUA repeatability | ±5% (Broadband Ultrasound Attenuation) |
| Calibration | Automatic with human body simulation module and temperature compensation |
| Dimensions | 330mm (W) × 360mm (H) × 645mm (L) |
| Weight | 13 kg (28.7 lbs) |
| Power supply | AC 220V ± 10%, 50Hz, 125W |
| Operating temperature | 10°C to 40°C (50°F to 104°F) |
| Operating humidity | 30% to 70% (non-condensing) |
| Operating system | Windows XP, 7, 8, 10 (32/64-bit compatible) |
| Connectivity | USB interface, Bluetooth wireless, PACS network compatible |
| Language support | Chinese and English with interface switching |
| Reference databases | Multi-ethnic (Asian, European, Middle Eastern, African) |
| Warranty | 2 Year Manufacturer Warranty |
Diagnostic parameters & outputs
| Parameter | Description |
|---|---|
| BUA | Broadband Ultrasound Attenuation — measures ultrasound energy loss through bone, correlating with bone density and microarchitecture quality |
| SOS | Speed of Sound — ultrasound velocity through bone tissue, reflecting bone density and elastic properties |
| OI | Osteo Index — composite bone mass index combining BUA and SOS for comprehensive bone quality assessment |
| T-Score | Comparison to peak bone mass of healthy young adults (Standard Deviation Units) |
| Z-Score | Comparison to age-matched reference population (Standard Deviation Units) |
| Adult Ratio | Patient’s bone density as percentage of young adult reference mean |
| Age Ratio | Patient’s bone density as percentage of age-matched reference mean |
| OPR | Osteo Profile Ratio — integrated bone health metric combining multiple ultrasound parameters |
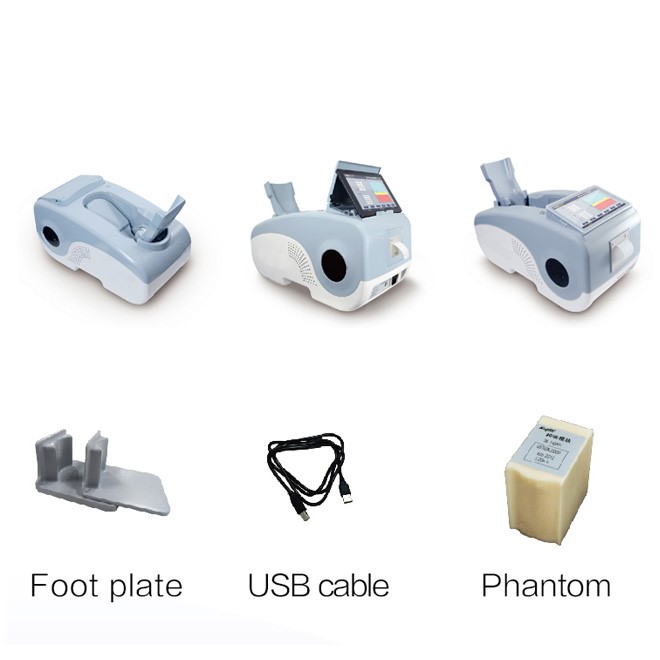
Standard instrument composition
| Item | Description | Qty |
|---|---|---|
| MDLBD05 Main Unit | Ultrasonic bone densitometer with integrated display and control panel | 1 |
| Oil Bladder Probe | Dry-type dual-transmission ultrasound transducer with permanent operational lifespan | 1 |
| Calibration Module | Human body simulation phantom for automatic system verification and quality control | 1 |
| USB Cable | High-speed data transfer interface for computer connectivity | 1 |
| Power Cord | AC 220V ± 10%, 50Hz power supply cable | 1 |
| Software Suite | Windows-compatible application with patient database, reporting, and connectivity modules | 1 |
| User Manual | Comprehensive operating instructions, quality control procedures, and troubleshooting guide | 1 |
| Quick Reference Guide | Laminated operator reference card for common procedures and measurements | 1 |
Get in touch for pricing & availability
Contact our sales team to discuss the MDLBD05 bone density scanner for your clinical facility. We provide comprehensive product demonstrations, customised quotations, and technical support for integration into your bone health assessment program.
Email: info@medicaldevicelab.com
Hours: Monday–Friday, 8 AM–6 PM PST | 24/7 Emergency Support Available








Reviews
There are no reviews yet.